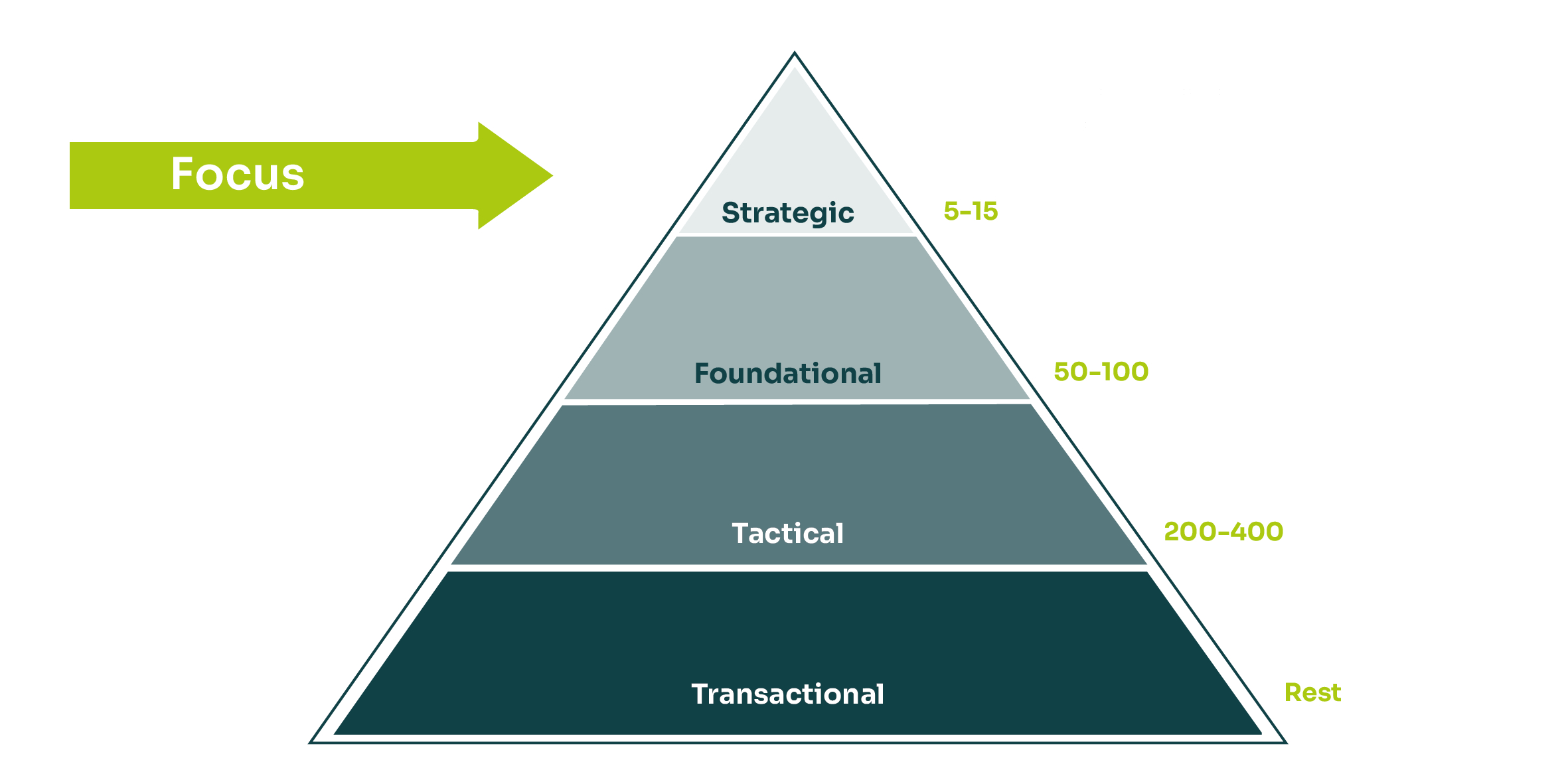
These constraints fundamentally change how sourcing decisions are made. Technology sits at the core of value creation and organizations depend heavily on specialized providers. Many partnerships are longterm by necessity, shaped by validation requirements, embedded platforms and high switching costs.
Price and performance alone no longer determine the right choice. Validation effort, documentation quality and audit readiness increasingly define total cost of ownership. Suppliers that integrate regulatory requirements into their delivery models often create structural advantages — even when their commercial offers appear less attractive at first glance.
In practice, fragmented supplier landscapes drive revalidation effort, slow down change cycles and dilute accountability during audits. Reducing interfaces and clarifying responsibility therefore becomes as important as reducing rates.